 
To calculate all possible term symbols for a given electron configuration the process is a bit longer. Its ground state term symbol is then Term symbols for an electron configuration As fluorine 2p subshell is more than half filled, J = L + S = 3/2. The first three electrons can take m s = 1/2 (↑) but the Pauli exclusion principle forces the next two to have m s = −1/2 (↓) because they go to already occupied orbitals.ģ. There are three orbitals ( m l = 1, 0, −1) that can hold up to 2(2 l+1) = 6 electrons. So we have five electrons to place in subshell p ( l = 1).Ģ. Discard the full subshells and keep the 2p 5 part. if the subshell is half-filled, then L will be 0, so J = S.Īs an example, in the case of fluorine, the electronic configuration is: 1s 22s 22p 5.ġ.if more than half-filled, take the maximum value J = L + S.if less than half of the subshell is occupied, take the minimum value J = | L − S |.The overall L is calculated by adding the m l values for each electron (so if there are two electrons in the same orbital, then we add twice that orbital's m l). That is the same as multiplying ½ times the number of unpaired electrons. The overall S is calculated by adding the m s values for each electron. 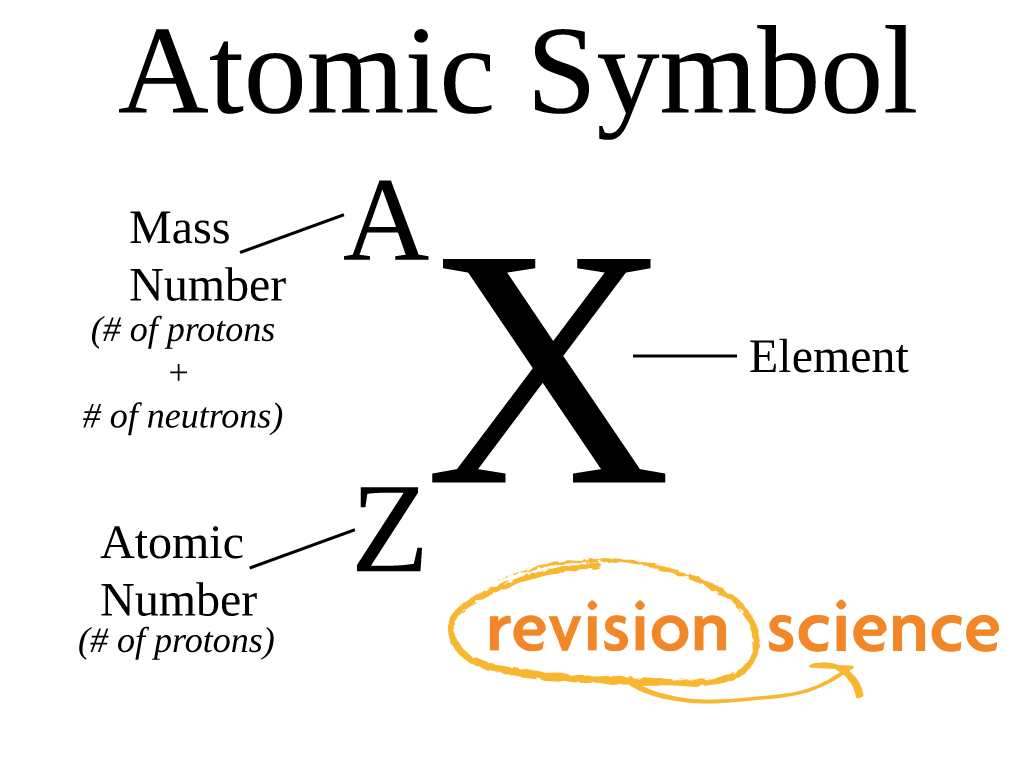
Once all orbitals in a subshell have one electron, add a second one (following the same order), assigning m s = −1/2 to them. First, we fill the orbitals with highest m l value with one electron each, and assign a maximal m s to them (i.e.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
